 Now, he put a negatively charged metal plate on one side of the cathode rays to go past the anode, and a positively charged metal plate on the other side. Then he conducted a Second experiment, to prove the charge carried by the cathode rays was negative or positive. From this, he deduced that the electric charge and the cathode rays must be combined and are the same entity. 
From the first experiment, he discovered that the electrometers stopped measuring electric charge. The metal had two small diversions(slits), leading to an electrometer that could measure a small electric charge. 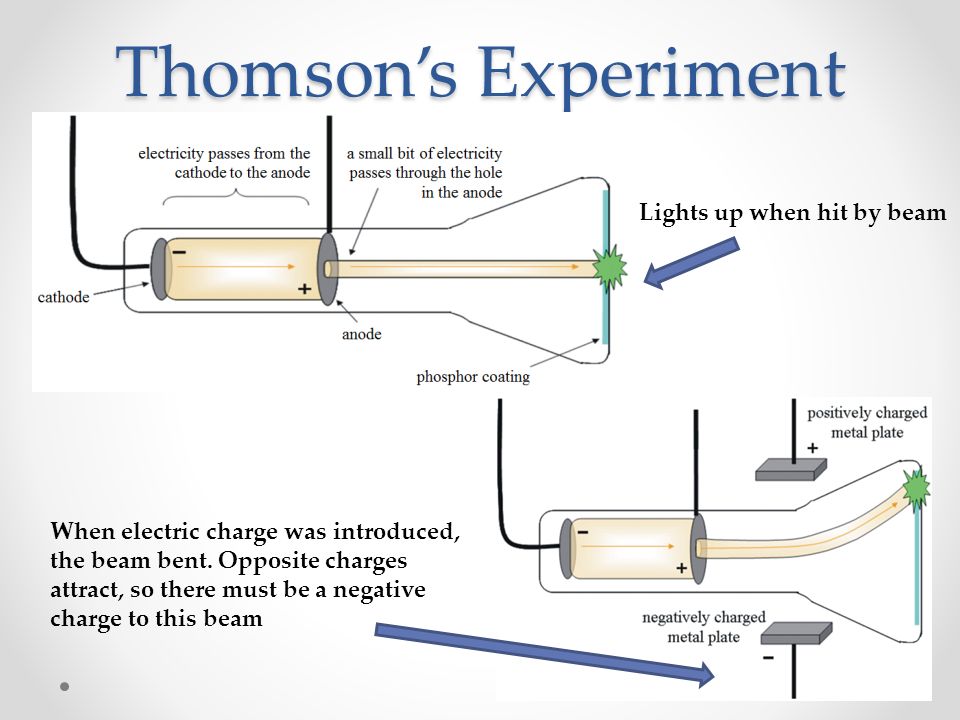
He built his cathode ray tube with a metal cylinder on the other end. Thompson, conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. Screen: - The inner layer of the screen is coated with phosphorus, and produces fluorescence when cathode rays hit the screen by a process of phosphorus excitation.Īquadag: - It is an aqueous solution of graphite used to collect the secondary emitted electrons which are required to keep the cathode ray in electrical equilibrium. The electron gun has a heater, cathode, pre-accelerating anode, focusing anode and accelerating anode.ĭeflecting Plates: - They produce a uniform electrostatic field only in one direction, and accelerate particles in only one direction. A wire is connected from anode to cathode to complete the electrical circuit.Įlectron Gun Assembly: - It is the source of the electron beams. When a potential difference is applied, the electrons jump to an excited state and travel at high speeds to jump back-and-forth inside the vacuum glass chamber and when some cathode rays certain molecules of the cathode screen, they emit light energy. Since electrons are repelled by the negative electrode, the cathode is the source of cathode rays inside a vacuum environment. 
The cathode is a negative electrode, Anode is the positive electrode. ACTION OF HIGH SPEED ELECTRONS ON METHANE, OXYGEN AND CARBON MONOXIDE.Cathode rays are a beam of negatively charged electrons traveling from the negative end of an electrode to the positive end within a vacuum, across a potential difference between the electrodes. THE ACTION OF HIGH-SPEED CATHODE RAYS ON THE SIMPLER ALCOHOLS, ALDEHYDES AND KETONES, AND ON ETHYLENE. Ozone hazards incurred by electron Van de Graaff operation. Human Factors: The Journal of the Human Factors and Ergonomics Society 1963, 5 Space Cabin Atmosphere Trace Contaminants and Their Possible Influence on Visual Parameters. Journal of Chemical & Engineering Data 2001, 46 Potentiometric Study of the Dissociation Quotients of Aqueous Dimethylammonium Ion As a Function of Temperature and Ionic Strength. Journal of Chemical & Engineering Data 2003, 48 
Determination of Stoichiometric Dissociation Constants of Acetic Acid in Aqueous Solutions Containing Acetic Acid, Sodium Acetate, and Sodium Chloride at (0 to 60) ☌. Journal of Chemical & Engineering Data 2005, 50 Re-evaluation of Stoichiometric Dissociation Constants from Electrochemical Cell Data for Formic Acid at Temperatures from (0 to 60) ☌ and for Some Other Aliphatic Carboxylic Acids at (18 or 25) ☌ in Aqueous Potassium Chloride Solutions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
